A) sp2
B) sp3
C) sp3d
D) sp3d2
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the predicted molecular geometry of the IBr3 molecule according to the VSEPR model?
A) tetrahedral
B) T-shaped
C) bent
D) trigonal planar
E) seesaw
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Atoms of period 3 and beyond can undergo sp3d2 hybridization,but atoms of period 2 cannot.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
The Lewis structure of formaldehyde,CH2O,is shown.Use VSEPR model to predict the molecular geometry and the H-C-H bond angle.Outline your reasoning. 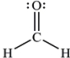
Correct Answer

verified
There are three electron groups around t...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Predict the molecular geometry and polarity of the SO2 molecule.
A) linear,polar
B) linear,nonpolar
C) bent,polar
D) bent,nonpolar
E) trigonal planar,polar
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the number of lone electron pairs on the central atom of a molecule having a seesaw molecular geometry,such as SF4?
A) 1
B) 2
C) 3
D) 4
E) 6
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hybridization of the central nitrogen atom in the molecule N2O is
A) sp.
B) sp2.
C) sp3.
D) sp3d.
E) sp3d2.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The angles between sp2 hybrid orbitals are 109.5°.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model,a molecule with the general formula AB4 with two lone pairs on the central atom will have a _____ molecular geometry.
A) tetrahedral
B) square pyramidal
C) square planar
D) octahedral
E) seesaw
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of XeO2F2 as predicted by the VSEPR model? 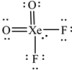
A) square planar
B) tetrahedral
C) square pyramidal
D) seesaw
E) octahedral
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
According to molecular orbital theory,all diatomic molecules with an even number of electrons are diamagnetic.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the VSEPR model,what is the predicted molecular geometry of the PCl3 molecule?
A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal
E) tetrahedral
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
To correctly determine the molecular shape of a molecule requires that you first draw the Lewis structure for the molecule.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the bond order for the species O2−?
A) 1
B) 1.5
C) 2
D) 2.5
E) None of the
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of HOF as predicted by the VSEPR model?
A) trigonal pyramidal
B) bent
C) tetrahedral
D) linear
E) trigonal planar
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which one of the following molecules is the indicated type of hybridization not appropriate for the central atom?
A) BeCl2; sp2
B) SiH4; sp3
C) BF3; sp2
D) C2H2; sp
E) H2O; sp3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule with the formula AB4 and a square planar molecular geometry uses _________ to form its σ bonds.
A) sp hybrid orbitals
B) sp2 hybrid orbitals
C) sp3 hybrid orbitals
D) sp3d hybrid orbitals
E) sp3d2 hybrid orbitals
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assume that an N2 molecule absorbs a photon and goes to its first excited state.
A) The excited N2 is less stable than N2 in the ground state.
B) The excited N2 is more stable than N2 in the ground state.
C) The bond length of N2 in the excited state is shorter than the bond length of N2 in the ground state.
D) The stability of the molecule is unchanged.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which molecule is the central atom sp2 hybridized?
A) SO2
B) N2O
C) BeCl2
D) NF3
E) PF5
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many π bonds are there in one molecule of 1,5-hexadiyne (shown below) ? 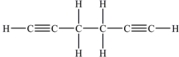
A) 2
B) 4
C) 6
D) 11
E) 15
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 137
Related Exams