A) Hemiacetal
B) Di-ether
C) Di-alkoxy alkane
D) Acetal
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name for the following compound? 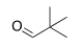
A) pivaldehyde
B) 2,2-dimethylpropanal
C) tert-butyl aldehyde
D) 2,2-dimethylpentanal
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most reactive carbonyl compound? 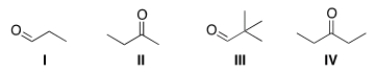
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Why are strongly acidic conditions not used in the formation of enamines and imines?
A) The carbonyl will be protonated.
B) The amine will be completely protonated.
C) The product is not stable to strong acid.
D) An enol will be formed.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 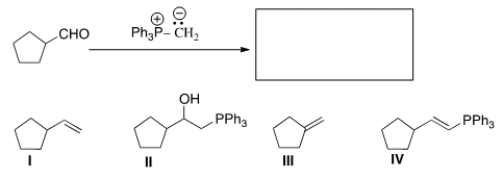
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 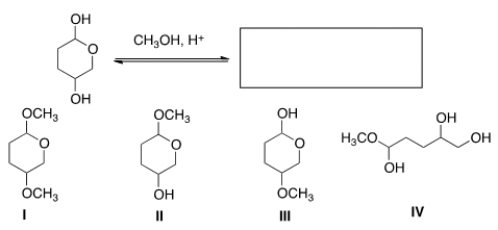
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the driving force for the Wittig reaction?
A) The formation of an alkene
B) The deprotonation of a phosphonium salt
C) The elimination of triphenylphosphine oxide
D) The formation of a phosphonium salt
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Butanal (MW= 72) has a boiling point of 76 °C while butanol (MW= 74) has a boiling point of 118 °C. What accounts for this difference in boiling points?
A) Butanol is polar while butanal is not polar.
B) Butanol can exhibit dipole-dipole interactions while butanal cannot.
C) Butanol can hydrogen bond and butanal cannot hydrogen bond.
D) Butanal is less sterically hindered than butanol.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC for the following compound? 
A) 1-formyl-2-nitropropane
B) 1-formyl-3-nitrobutane
C) 2-nitrobutanal
D) 3-nitrobutanal
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What needs to be done to make the following reaction go to starting materials? 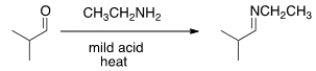
A) Heat the reaction.
B) Add aqueous acid.
C) Add aqueous base.
D) Add water.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 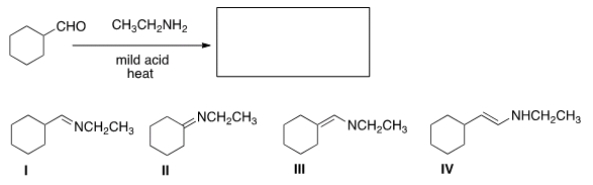
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What is (are) the product(s) of the following reaction? 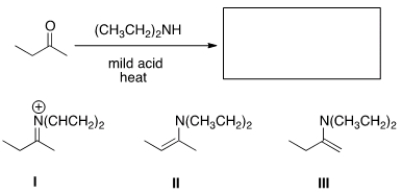
A) I only
B) II only
C) III only
D) II and III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cyclic acetals are used as protecting groups for ketones or aldehydes because they are inert to all of the following reagents except
A) aqueous acid.
B) aqueous base.
C) oxidizing reagents.
D) reducing reagents.
F) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What needs to be done to make the following reaction proceed? 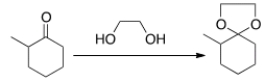
A) Heat the reaction.
B) Add an acid catalyst only.
C) Add a base catalyst only.
D) Heat the reaction and add an acid catalyst.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 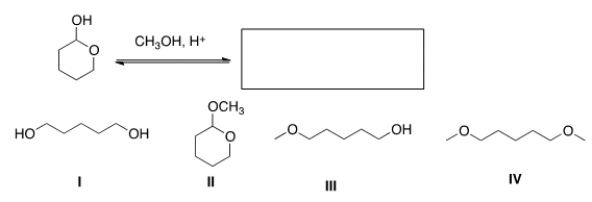
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 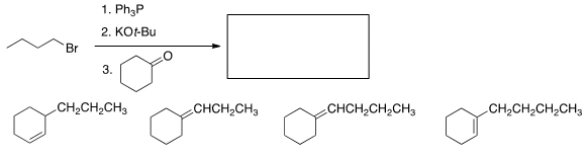
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Is the following reaction reversible and, if so, under what conditions? 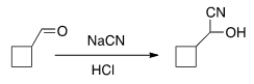
A) No
B) Yes, under acidic conditions
C) Yes, using Pd/C
D) Yes, under basic conditions
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below? 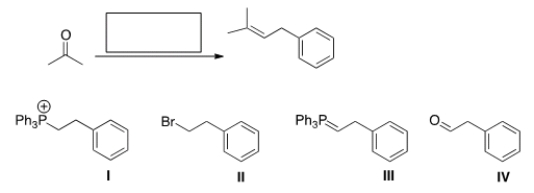
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using 1H NMR spectroscopy, how can you tell the difference between an aldehyde and a ketone?
A) An aldehyde has a C-H stretch (one or two) between 2700-2830 cm-1.
B) An aldehyde has a proton signal between 9-10 ppm.
C) A ketone has signals around 2-3 ppm.
D) A ketone has a signal around 200 ppm.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 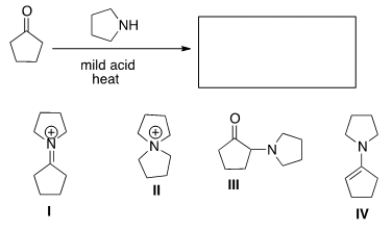
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 45
Related Exams