A) Amide, aromatic, ether, ketone.
B) Amide, aromatic, ester, ketone.
C) Amine, aromatic, ester, ketone.
D) Amine, aromatic, ether, ketone.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What intermolecular force is generally considered the weakest?
A) Hydrogen bonding
B) London dispersion forces
C) Dipole-dipole
D) Ion-ion
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly matches the molecules to the names of the functional groups? I. CH3OCH3 Ether II) CH3CONH2 Amine III) CH3SH Thiol IV) CH3CHO Alcohol
A) I and II
B) II and III
C) III and IV
D) I and III
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules contain the same functional groups?
A) I, II, IV
B) I, II, III
C) II, III, IV
D) I, III, IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following lists contains common heteroatoms found in organic molecules?
A) N, O, S, P, Cl
B) Na, O, S, P, Cl
C) Na, Mg, S, N, Cl
D) Na, Mg, O, N, Cl
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures contains an amide? 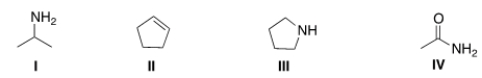
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkanes is expected to have the highest melting point? 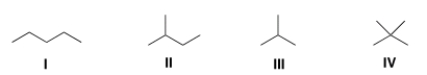
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is expected to be H2O soluble? 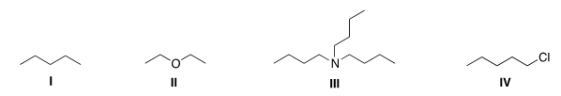
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing melting point, putting the compound with the highest melting point first. 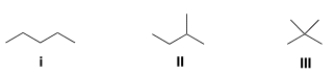
A) I > II > III
B) II > III > I
C) III > II > I
D) III > I > II
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do heteroatoms confer reactivity on a particular molecule?
A) Because they have lone pairs and create electron-rich sites on carbon.
B) Because they have lone pairs and create electron-deficient sites on carbon.
C) Because they are electronegative and act as electrophiles.
D) Because they are electropositive and act as nucleophiles.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicated bond is: 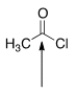
A) Nucleophilic because it is electron-deficient.
B) Electrophilic because it is electron-rich.
C) Nucleophilic because it is electron-rich.
D) Electrophilic because it is electron-deficient.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the lowest boiling point? 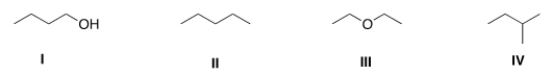
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly matches the molecules to the names of the functional groups? I. CH3OH Carboxylic acid II) CH3CO2CH3 Ester III) CH3COCH3 Ketone IV) H2CO Alcohol
A) I and II
B) III and IV
C) II and III
D) II and IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do bonds confer reactivity on a particular molecule?
A) Because bonds are difficult to break in chemical reactions.
B) Because bonds make a molecule an acid.
C) Because bonds are easily broken in chemical reactions.
D) Because bonds make a molecule an electrophile.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicated bond is: 
A) Nucleophilic because it is electron-deficient.
B) Electrophilic because it is electron-deficient.
C) Nucleophilic because it electron-rich.
D) Electrophilic because it is electron-rich.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about vitamin C, drawn below, are true? 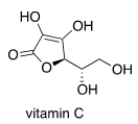
A) Vitamin C is insoluble in H2O.
B) Vitamin C is soluble in H2O.
C) Vitamin C is an aliphatic hydrocarbon.
D) Vitamin C contains a ketone functional group.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds can form intermolecular hydrogen bonds with a molecule similar to itself? 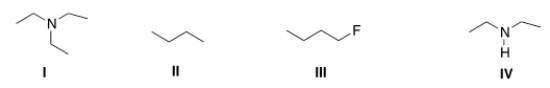
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 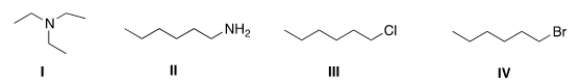
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds would be expected to be more soluble in hexane (C6H14) ? 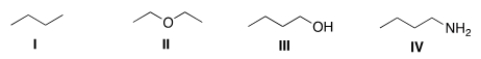
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following intermolecular forces would not form between similar molecules of the structure below? 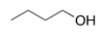
A) London dispersion forces
B) Ion-ion
C) Hydrogen bonding
D) Dipole-dipole
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 48
Related Exams