Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element whose Lewis symbol is correct.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The lattice energy of rubidium chloride is the energy change accompanying the process
Rb+(g) + Cl-(g) RbCl(s)
Calculate the lattice energy of RbCl using the following data: 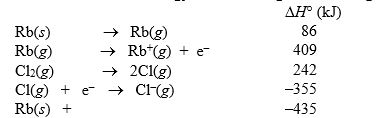
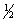 Cl2(g) RbCl(s)
Cl2(g) RbCl(s)
Correct Answer

verified
Lattice en...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Arrange calcium, rubidium, sulfur, and arsenic in order of decreasing electronegativity.
A) S > As > Rb > Ca
B) S > As > Ca > Rb
C) As > S > Rb > Ca
D) As > S > Ca > Rb
E) None of the above orders is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an ionic compound?
A) H2S
B) NH3
C) I2
D) KI
E) CCl4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which of the following elements (in their normal, stable, forms) would it be correct to describe the bonding as involving "electron pooling"?
A) hydrogen
B) helium
C) sulfur
D) iodine
E) aluminum
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in order of increasing bond strength.
A) C-I < C-Br < C-Cl < C-F
B) C-F < C-Cl < C-Br < C-I
C) C-Br < C-I < C-Cl < C-F
D) C-I < C-Br < C-F< C-Cl
E) None of the above orders is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the bond energies provided below, calculate H° for the reaction CH4(g) + 4Cl2(g) CCl4(g) + 4HCl(g) Bond energies: C-H = 413 kJ/mol, Cl-Cl = 243 kJ/mol, C-Cl = 339 kJ/mol, H-Cl = 427 kJ/mol
A) 1422 kJ
B) 440 kJ
C) 110 kJ
D) -110 kJ
E) - 440 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The diameter of a chloride ion is 362 pm, and the diameter of a potassium ion is 276 pm. What is the distance between the nuclei of adjacent chloride and potassium ions in solid potassium chloride?
A) 1276 pm
B) 638 pm
C) 319 pm
D) 181 pm
E) 138 pm
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The majority of elements are good electrical conductors when in solid form.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of these substances are the atoms held together by metallic bonding?
A) CO2
B) Si
C) Br2
D) S8
E) Cr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Analysis of an unknown substance showed that it has a high boiling point and is brittle. It is an insulator as a solid but conducts electricity when melted. Which of the following substances would have those characteristics?
A) HCl
B) Al
C) KBr
D) SiF4
E) I2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The electrostatic energy of two charged particles is inversely proportional to the square of the distance between them.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange aluminum, nitrogen, phosphorus, and indium in order of increasing electronegativity.
A) Al < In < N < P
B) Al < In < P < N
C) In < Al < P < N
D) In < P < Al < N
E) None of the above orders is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Bond energy increases as bond order increases, for bonding between a given pair of atoms.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Combustion of a fat will release more energy than combustion of an equal mass of carbohydrate because
A) fats contain more bonds to oxygen than carbohydrates.
B) fats contain fewer bonds to oxygen than carbohydrates.
C) the total energy of the carbon-carbon and carbon-hydrogen bonds in fats is greater than the energy content of the carbon-oxygen and oxygen-hydrogen bonds in the reaction products (carbon dioxide and water) .
D) the total energy of the carbon-carbon and carbon-hydrogen bonds in fats is greater than the energy content of the bonds in carbohydrates.
E) fats have higher molar masses than carbohydrates.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of these substances are the atoms held together by polar covalent bonding?
A) SrCl2
B) CsCl
C) ClF
D) TiF2
E) S8
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lattice energy of CaF2 is the energy change for which one, if any, of the following processes?
A) Ca2+(s) + 2F-(g) CaF2(g)
B) CaF2(g) CaF2(s)
C) Ca(g) + 2F(g) CaF2(s)
D) CaF2(aq) CaF2(s)
E) none of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct formula for a compound formed from barium and nitrogen.
A) BaN
B) BaN2
C) Ba2N3
D) Ba2N
E) Ba3N2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Covalently bonded substances do not necessarily exist as separate molecules.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 74
Related Exams