A) carbon (coke) .
B) carbon monoxide.
C) molecular hydrogen.
D) sodium.
E) electrolysis.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Cast iron as it is prepared in a blast furnace is a product of high purity.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The chemical formula of magnetite can be expressed as either Fe3O4 or FeO Fe2O3
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The following two reactions represent the chemical reactions that can take place when magnesium is burned in air.2Mg(s) + O2(g) 2MgO(s) and 3Mg(s) + N2(g) Mg3N2(s)
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following diagrams represents an insulator 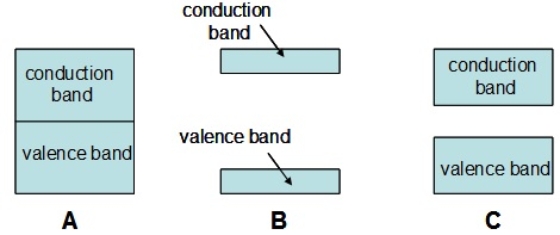
A) A
B) B
C) C
E) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Hall process, ____________ is reduced ____________.
A) nickel; electrolytically
B) aluminum; electrolytically
C) nickel; by reaction with metallic sodium
D) aluminum; by reaction with metallic sodium
E) copper; electrolytically
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the chemical formula of pyrite.
A) FeS
B) Fe2S3
C) FeS2
D) Fe3S2
E) None of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The alkali metals listed in order of increasing melting point is consistent with which of the following
A) Li < Na < K < Rb < Cs
B) Cs < Rb < K < Na < Li
C) Be < Mg < Ca < Sr < Ba
D) Ba < Sr < Ca < Mg < Be
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Alloys of iron that contain 1.0-1.5% carbon and some manganese, phosphorus, silicon, and sulfur are called
A) steel.
B) cast iron.
C) coke.
D) pig iron.
E) hematite.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction 3Fe(s) + C(s) Fe3C(s) , H = 21 kJ/mol and S = 20.4 J/mol·K at 25 C. Estimate the minimum temperature above which the formation of cementite (Fe3C) is favored.
A) 1.0 C
B) 970 C
C) 700 C
D) 1000 C
E) 760 C
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Potassium superoxide, KO2(s), is used in the breathing systems of biohazard suits. The following separate chemical equations show how KO2(s) reacts with moisture and carbon dioxide in exhaled air to generate oxygen gas. 2KO2(s) + 2H2O(l) 2KOH(aq) + O2(g) + H2O2(aq) 4KO2(s) + 2CO2(g) 2K2CO3(s) + 3O2(g)
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The flotation process used in metallurgy involves
A) the roasting of sulfides.
B) separation of gangue from ore.
C) electrolytic reduction.
D) chemical reduction of a metal.
E) zone refining.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these ions is most likely to substitute for Ca2+ in the human body
A) Cl-
B) Sr2+
C) K+
D) S2-
E) Pb2+
G) A) and E)
Correct Answer

verified
B
Correct Answer
verified
True/False
The following equation correctly represents the balanced chemical equation illustrating the thermite reaction.2Al(s) + Fe2O3(s) Al2O3(s) + 2Fe(l)
B) False
Correct Answer

verified
True
Correct Answer
verified
Multiple Choice
Write the chemical formula of corundum.
A) Al3O2
B) Al2O3
C) AlO3
D) Al3O
E) None of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barite, anglesite, and epsomite are all examples of what type of mineral
A) carbonates
B) oxides
C) sulfides
D) sulfates
E) silicates
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is used to produce chromium metal at high temperature.Cr2O3(s) + 2Al(s) 2Cr(l) + Al2O3(s) Which element is oxidized and which is reduced
A) Both chromium and aluminum are oxidized
B) Chromium is oxidized and aluminum is reduced
C) Chromium is reduced and aluminum is oxidized
D) Neither chromium or aluminum are oxidized
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these metals would normally be obtained by electrolytic reduction
A) aluminum
B) chromium
C) copper
D) iron
E) zinc
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum is an active metal, but does not corrode as iron does because
A) Al does not react with O2.
B) a protective layer of Al2O3 forms on the metal surface.
C) Al is harder to oxidize than is Fe.
D) the enthalpy of formation of aluminum oxide is negative.
E) aluminum has a high tensile strength.
G) C) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
In the production of potassium metal, the source of electrons in the reduction of K+ ions is
A) H2(g)
B) Na(g)
C) CO(g)
D) CaO(s)
E) electrolysis
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 63
Related Exams