A) HCl, NaCl
B) HNO3, NaNO3
C) H3PO4, NaH2PO4
D) H2SO4, CH3COOH
E) NH3, NaOH
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.30 M HClO was titrated with 0.30 M NaOH. The following data were collected during the titration. Determine to Ka for HClO. 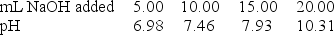
A) 1.1 × 10-7
B) 3.5 × 10-8
C) 1.2 × 10-8
D) 4.9 × 10-11
E) None of the answers is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which indicator would be the best to use if 0.050 M benzoic acid (Ka = 6.6 × 10-5) is titrated with 0.050 M NaOH?
A) bromphenol blue, pH range: 3.0 - 4.5
B) bromcresol green, pH range: 3.8 - 5.4
C) alizarin, pH range: 5.7 - 7.2
D) phenol red, pH range: 6.9 - 8.5
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n) ________ is an ion containing a central metal cation bonded to one or more molecules or ions.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is correct?
A) Solubility is the concentration of unsaturated ions in solution.
B) Ksp is the same as the solubility.
C) Ksp is an equilibrium constant and solubility is the concentration of a saturated solution.
D) Ksp is the solubility product constant for a precipitate.
E) Solubility = Kw × Ksp.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a buffer that consists of 0.45 M CH3COOH and 0.35 M CH3COONa? [Ka(CH3COOH) = 1.8 × 10-5]
A) 3.43
B) 4.64
C) 4.85
D) 4.74
E) 6.06
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a strong acid is titrated with a strong base, the pH at the equivalence point
A) is greater than 7.0.
B) is equal to 7.0.
C) is between 3.5 and 7.0.
D) is equal to the pKa of the acid.
E) is equal to 3.5.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
An indicator is a weak acid that is one color in its protonated form and another color in its deprotonated form.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which combination may be used to prepare a buffer having a pH of 8.8? Ka= 7 × 10-3 for H3PO4; 8 × 10-8 for H2PO4-; 5 × 10-13 for HPO42-
A) NaH2PO4 only
B) NaH2PO4 and Na2HPO4
C) Na3PO4 and Na2HPO4
D) NaHPO4 and H3PO4
E) H3PO4 and Na3PO4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A wildlife biologist is interested in testing the pH of the water in a lake. He obtains a 200.0-mL sample of the lake water and titrates this sample with a 0.050 M NaOH solution. Neutralization of the lake water requires 40.0 mL of the NaOH solution. Assuming that lake water sample only contains a strong acid, estimate the pH of the lake.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is more soluble in a basic solution than in pure water?
A) Mg(OH) 2
B) CaHPO4
C) NaCl
D) CaCO3
E) AgI
G) B) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
For PbCl2 (Ksp = 2.4 × 10-4) , will a precipitate of PbCl2 form when 0.10 L of 3.0 × 10-2M Pb(NO3) 2 is added to 400 mL of 9.0 × 10-2M NaCl?
A) Yes, because Q > Ksp.
B) No, because Q < Ksp.
C) No, because Q = Ksp.
D) Yes, because Q < Ksp.
F) B) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
The molar solubility of tin(II) iodide is 1.28 × 10-2 mol/L. What is Ksp for this compound?
A) 8.39 × 10-6
B) 1.28 × 10-2
C) 4.20 × 10-6
D) 1.64 × 10-4
E) 2.10 × 10-6
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is appreciably more soluble in 1 M HNO3 than in pure water?
A) FeCO3
B) AgBr
C) BaSO4
D) NaNO3
E) NaCl
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the effect on the equilibrium when sodium formate is added to a solution of formic acid? HCOOH(aq)  H+(aq) + HCOO-(aq)
H+(aq) + HCOO-(aq)
A) There is no change in the equilibrium.
B) The equilibrium shifts to the right.
C) More information is needed to answer the question.
D) The equilibrium shifts to the left.
E) The pH decreases.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Bromothymol blue is a common acid-base indicator. It has a Ka equal to 1.6 × 10-7. Its un-ionized form is yellow and its conjugate base is blue. What color would a solution have at pH = 5.8?
Correct Answer

verified
Correct Answer
verified
True/False
The endpoint in a titration is defined as the point when the appropriate indicator changes color.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An acetic acid buffer containing 0.50 M CH3COOH and 0.50 M CH3COONa has a pH of 4.74. What will the pH be after 0.0020 mol of HCl has been added to 100.0 mL of the buffer?
A) 4.77
B) 4.71
C) 4.68
D) 4.62
E) 1.70.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 10.0-mL sample of 0.75 M CH3CH2COOH is titrated with 0.30 M NaOH. What is the pH of the solution after 22.0 mL of NaOH have been added to the acid? [Ka(CH3CH2COOH) = 1.3 × 10-5]
A) 5.75
B) 4.94
C) 4.83
D) 4.02
E) 3.95
G) D) and E)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Methyl red is a common acid-base indicator. It has a Ka equal to 6.3 × 10-6. Its un-ionized form is red and its anionic form is yellow. What color would a methyl red solution have at pH = 7.8?
A) green
B) red
C) blue
D) yellow
E) violet
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 133
Related Exams