A) The solution should change from yellow to blue.
B) The solution should change from blue to yellow.
C) The solution should not change color.
D) The solution should become colorless because H2O is a colorless liquid.
E) It is impossible to tell without performing a measurement.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the label used for an isomer which is dextrorotatory?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the appropriate energy-level diagram for the octahedral coordination complex hexaammineiron(III) ?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ ________ have the ability to hold the metal ion, in the complex ion, like a claw.
Correct Answer

verified
Correct Answer
verified
Short Answer
If the plane of polarization is rotated to the left then the isomer is ________.
Correct Answer

verified
Correct Answer
verified
Short Answer
d and l isomers of a chiral substance are called ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the systematic name for [CoCl3(H2O) ]-?
A) cobalt(III) chloride monohydrate
B) aquatrichlorocobalt(II)
C) aquatrichlorocobaltate(II)
D) aquatrichlorocobaltite(I)
E) monoaquotris(chloro) cobalt(IV)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 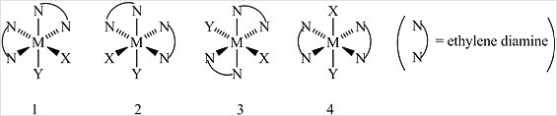 Which one of the following statements about these structures is incorrect?
Which one of the following statements about these structures is incorrect?
A) Structures 1 and 2 are optical isomers.
B) Structures 1 and 3 are optical isomers.
C) Structures 1 and 3 are different complexes.
D) Structures 1 and 4 are geometrical isomers.
E) Structures 3 and 4 are the same complex.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions is most likely to form colored compounds?
A) Sc3+
B) Cu+
C) Zn2+
D) Cr3+
E) Ca2+
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the appropriate energy-level diagram for the tetrahedral coordination complex tetrachloromanganate(II) ?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What geometry is particularly common for complexes of d10 metal ions?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are phosphates being eliminated from detergents?
A) Detergents with phosphates are less efficient cleansers.
B) By chelating Ca2+ ions, the phosphates allow more efficient cleansing.
C) Phosphates are important plant nutrients which increase algae growth and deplete oxygen.
D) None of these choices.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 132 of 132
Related Exams