A) (1) HBr, H2O2; (2) KOC(CH3) 3 ; (3) MCPBA; (4) CH2=CPh2
B) (1) NBS, hn ; (2) KOC(CH3) 3 ; (3) CHBr3, KO(CH3) 3 ; (4) a.LiCuPh2, b.H2O
C) (1) H2SO4(aq.) , D ; (2) MCPBA; (3) CHBr3, KO(CH3) 3 ; (4) CH2=CPh2
D) (1) Br2, FeBr3 ; (2) MCPBA; (3) CHBr3, KO(CH3) 3 ; (4) Ph2COCl
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the major organic product of the following reaction. 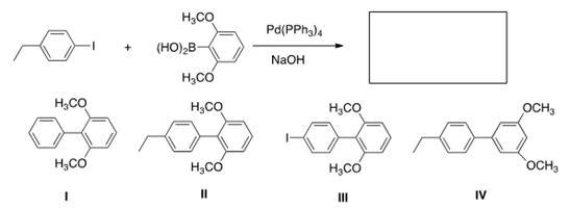
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the structure of the organic product Y formed in the following reaction sequence. 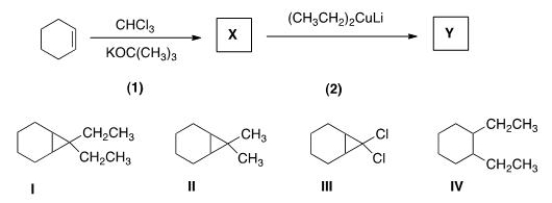
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the major organic product of the following reaction. 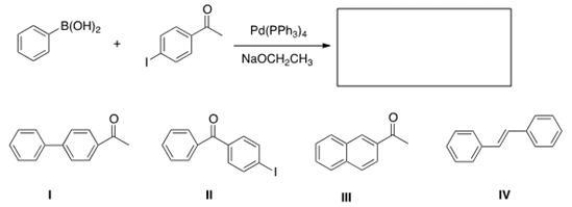
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would be the major organic product of the following Heck reaction?
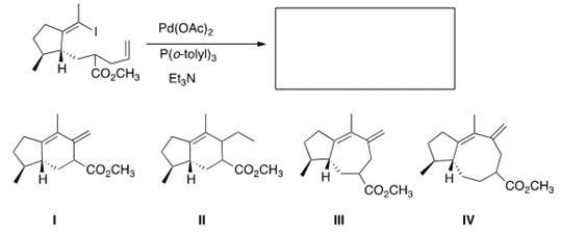
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following descriptions does not apply to methylene?
A) Methylene is sp2 hybridized.
B) Methylene is a neutral, reactive intermediate.
C) Methylene is a radical intermediate.
D) The formula of methylene is :CH2.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which class of compounds listed below does not react with organocuprate reagents to form a coupling product that contains a new carbon-carbon bond?
A) 1° Alkyl halides
B) 3° Alkyl halides
C) Vinyl halides
D) Aryl halides
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What do the Suzuki reaction, the Heck reaction, and the organocuprate reaction all have in common when they react with an alkyl halide?
A) All reactions form new carbon-carbon bonds.
B) They all use palladium as a catalyst in one step of the reaction.
C) They are all stereospecific reactions.
D) They all require harsh conditions.
F) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Identify the structure of the major organic product that results from the following reaction. 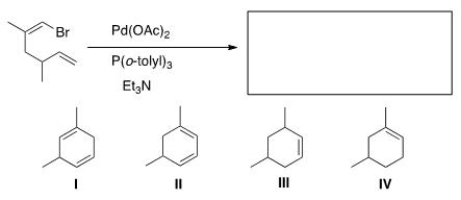
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly describes a reaction yielding a dichorocarbene?
A) The reaction of chloroform with KOC(CH3) 3.
B) The reaction of chloroform with Zn(Cu) .
C) The reaction of chloroform with (CH3) 2CuLi.
D) The reaction of diazomethane with KOC(CH3) 3.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the statement below that is not true about the Suzuki reaction.
A) The product of the Suzuki reaction is completely stereospecific.
B) The Suzuki reaction involves both an organoborane reagent and an organopalladium catalyst.
C) The Suzuki reaction forms more highly substituted alkenes.
D) The Suzuki reaction involves an oxidative addition followed by a reductive elimination.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Many of the reactions studied in this chapter are stereospecific.Why are stereospecific reactions important?
A) When a reaction produces a mixture of enantiomers, it is often very difficult to separate them.A mixture of products is often not useful.
B) Often stereoisomers of a particular compound will have very different biological effects on an organism.Only one isomer is biologically helpful, and the other may be harmful.
C) Often only one stereoisomer is biologically active, and coupling reactions are often used for the production of biological materials.
D) All of the choices are true.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As shown below, when cis-2-butene reacts with dichlorocarbene, only the cis-1,1-dichloro-2,3-dimethylcyclopropane is formed.What can we conclude about the nature of the reaction mechanism?
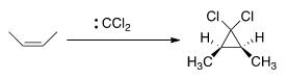
A) The mechanism is an SN1 mechanism.
B) The mechanism is a concerted.
C) The mechanism proceeds through a radical intermediate.
D) The mechanism is an E2 mechanism.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction?
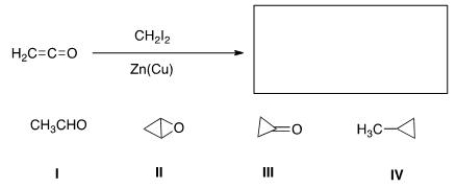
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction?
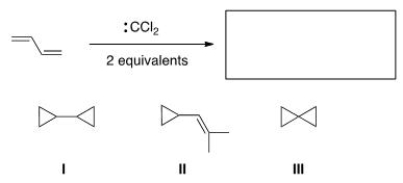
A) I
B) II
C) III
D) None of the choices is correct.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the appropriate sequence of starting material and reagents that will produce the target molecule shown below?
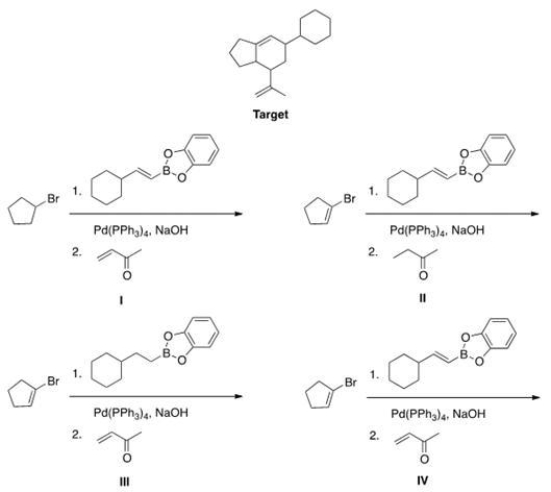
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the structure of the major organic product that results from the following reaction. 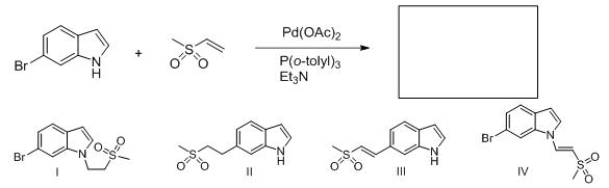
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Coupling reactions with vinyl halides are stereospecific.Which choice below best describes the expected product when trans-1-bromo-1-hexene reacts with (CH3) 2CuLi?
A) The reaction will only yield a trans-alkene.
B) The reaction will only yield a cis-alkene.
C) The reaction will only yield one enantiomeric product with R configuration.
D) The reaction will only yield one enantiomeric product with S configuration.
F) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Dichlorocarbene reacts with an alkene to form a cyclopropane derivative.In this reaction the dichlorocarbene acts as a(n)
A) Lewis base.
B) electrophile.
C) nucleophile.
D) Brønsted-Lowry base.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the reactions listed below is not stereospecific?
A) Suzuki reaction
B) Organocuprate coupling reaction
C) Heck reaction
D) Simmons-Smith reaction
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 37
Related Exams